Propecia (Finasteride) Description
Propecia belongs to the group of medications designed for treatment of benign prostatic hyperplasia (BPH) and male pattern baldness. This remedy is a competitive and specific inhibitor of 5-alpha-reductase of 2-type which is an endocellular enzyme turning into a more active androgen called dihydrotestosterone (DHT). It represents a synthetic 4-azasteroid composition. It decreases DHT level considerably either in blood or in gland tissue. The active substance is finasteride (brand names are Proscar or Propecia).
Propecia Application
Benign prostatic hyperplasia
In medical practice, Propecia is used for Benign Prostatic Hyperplasia (get to know more about this disease: https://www.mayoclinic.org/diseases-conditions/benign-prostatic-hyperplasia/symptoms-causes/syc-20370087) therapy known as ‘enlargement of prostatic gland’. The dosage of the remedy approved by Food and Drug Association is 5mg once a day. In order to achieve therapeutic results, six or more months of Propecia therapy may be required. Upon termination of medication’s administration, the disease may develop within the course of 6-8 months. Propecia decreases symptoms connected with prostate adenoma, such as difficult urination, nocturia, urine detain in the beginning of urination and weak urine stream.
Propecia for baldness
Within the course of five-year research of men with mild and moderate baldness degree, it was discovered that two out of three men taking 1mg of Propecia restored their hair growth. And on the contrary, those who didn’t use Propecia experienced hair loss. The same research study based on photos examined by peer review team of dermatologists has shown that 48 per cent of people taking Propecia had visible aftergrowth of hair, and other 42 per cent didn’t suffer from further hair loss any more.
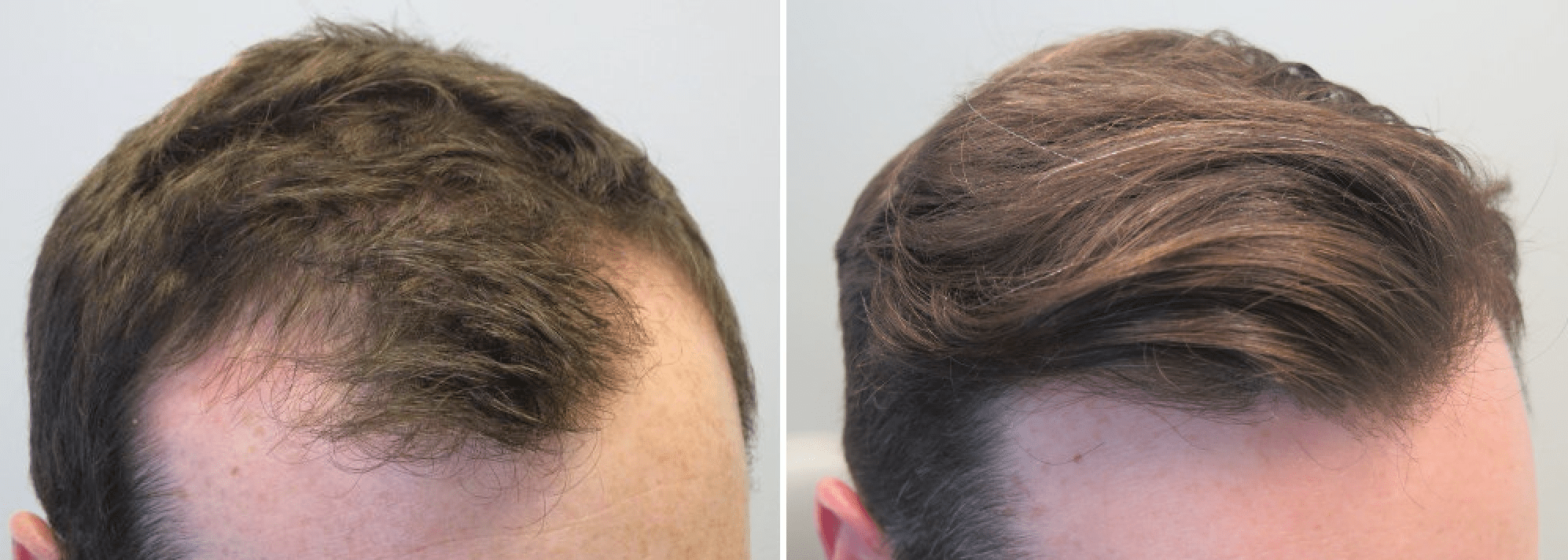
The average hair amount in therapy group remained higher of benchmark, and difference in hair quantity between participants of Propecia and placebo groups kept steadily growing within all five years of research. Propecia is effective only during the time of its administration. Hair grown during therapy start shedding during 6-12 month after therapy course termination. Clinical trials have shown that Propecia and Minoxidil take impact on both: parietal region and hairline, but it proves to be the most effective on parietal area.
Recent ten-year research study of 118 men suffering from alopecia (baldness) taking Propecia in dose of 1mg a day has demonstrated that 86 per cent of men who continued therapy during 10 years have experienced increase or preservation of hair growth rate, whereas only 14 per cent reported of further hair loss. It was stated that patients demonstrating higher hair growth rate during the first year of therapy had the most pronounced hair growth in five years of treatment. Moreover, almost 69 per cent of such patients experienced continuation of hair growth. Anyway, many patients who didn’t notice any growth within the first year of therapy has further improvements.
It was also stated that patients above 30 years of age generally has increased hair growth in perspective which is likely connected with the fact that such patients experience a large hair loss during their lifetime in comparison with other groups of population. Side-effects were observed only in 5,8 per cent of patients. Moreover, none of them reported such symptoms as depression or gynecomastia. Researchers concluded that Propecia efficiency in therapy of androgenetic alopecia wouldn’t disappear with time even in elderly patients (including those who are over forty years of age) and that the remedy had favorable tolerability.
The clinical trial has also examined patients continuing usage of the medication. The recent case–control study with participation of men suffering from hair loss and experiencing psychic complications after termination of Propecia intake has reported presence of depressive symptomatology and suicidal thoughts among patients who also had sexual side-effects during the medicine administration. Some consumers in an effort to save money purchase Proscar (finasteride 5mg) instead of Propecia and divide the tablet into several parts which draws the dosage closer to Propecia dose. In order to prevent from contact with active ingredient, tablets are covered with special coating. Dust and crumbs of broken tablets Proscar should be kept away from pregnant women or women who may become pregnant.
Off-label purposes
Due to its antiandrogenic features, Propecia is sometimes used in hormonal replacement therapy for transgender men in combination with estrogen form. Nevertheless, there are few clinical trials conducted for this purpose, and proofs of its efficiency are restricted. Sure enough, finasteride is a considerably weaker substance than such antiandrogens as spironolactone and cyprotoerone acetate.
Besides, its application is connected with high risk of depression and anxiety development either in men or in women; transgender people are in the group of particularly high risk, since such symptoms are spread among this group of patients. Therefore, Propecia prescription is not recommended in the capacity of antiandrogen for transgender men, since the medication increases risks of harmful emotional side-effects. It was discovered that Propecia promotes softening of clearance consequences after chronic alcohol misuse.
Propecia Mode of Action
This is a synthetic 4-azasteroid composition and competitive and specific inhibitor of 5-alpha-reductase of 2-type. This intracellular enzyme turns testosterone into active dihydrotestosterone. Inhibition of dihydrotestosterone production leads to reduction of prostatic glans in size. Propecia doesn’t have affinity to androgenic receptor due to which testosterone-dependent symptoms occur during its application in the form of weight gain and hirsuties. Resulting from Propecia application, DHT concentration in blood serum, urine and DHT in prostatic gland decreases causing reduction of prostatic gland in size, increase of maximal volume rate of urine stream, decrease of risk of acute urine detain and operative treatment necessity.
DHT concentration decrease in head skin prevents from miniaturization of hair follicles blocking process of boldness. After peroral intake, bioaccessibility makes 80 per cent; food intake doesn’t influence absorption. Propecia interacts with plasmatic proteins by 93 per cent and penetrates through hematoencephalic barrier. Metabolism occurs in liver under the influence of isoenzyme CYP3 A4 of P450 cytochrome. Around 60 per cent of medication gets cleared from organism with feces, around 40 per cent is cleared with urine. Terminal elimination half-life is five-six hours, whereas in patients over 70 years of age it makes eight hours. The effect lying in decrease of DHT concentration remains active within 24 hours.
Propecia Side-Effects
Propecia side-effects include: impotency (1.1-18.5%), premature ejaculation (7.2%), ejaculate volume decrease (0.9-2.8%), impairment of sexual function (2.5%), gynecomastia (2.2%), erectile dysfunction (1.3%), impairment of ejaculation (1.2%), pains in testicles. According to the product’s instruction, men who terminated Propecia therapy because of occurred side-effects experienced restoration like in men who continued the treatment further. Manufacturer also reports of cases of preserved erectile dysfunction, in spite of Propecia therapy termination. In December, 2010 depression was added to list of finasteride side-effects.
Contraindications
- Hypersensitivity to any of medication’s components;
- Women and children are not allowed taking Propecia (due to its capacity to turn testosterone into dihydrotestosterone, this medicine may cause abnormalities in sexual organs development of male fetus in pregnant women.
Condition of patients with big postvoid residual urine volume should be controlled thoroughly with respect to uropathy development caused by impenetrability of urinary tracts.
Interaction with Other Drugs
No clinically significant interactions with other remedies are detected.
Canadian Health&Care Mall Experts – https://canadianhealthcaremallrx.com